Nanografi’s CVD Graphene: A Breakthrough in Graphene Production
Graphene, often referred to as a “wonder material,” has revolutionized industries ranging from electronics to energy storage due to its remarkable properties. This two-dimensional carbon structure—a single layer of carbon atoms arranged in a honeycomb lattice—boasts exceptional electrical conductivity, mechanical strength, and thermal properties. One of the most promising methods for producing high-quality graphene is Chemical Vapor Deposition (CVD), a process widely used in Nanografi’s graphene production.
This article explores Nanografi’s CVD graphene, its synthesis, properties, and the vast potential it holds for various industries. We’ll also dive into how Nanografi stands at the forefront of graphene innovation, utilizing CVD technology to deliver high-quality graphene that meets the demands of the modern world.
What is CVD (Chemical Vapor Deposition) Graphene?
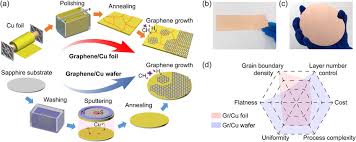
Chemical Vapor Deposition (CVD) is one of the most popular and reliable methods for producing high-quality graphene. In this technique, carbon atoms are introduced to a substrate (usually copper or nickel) via gaseous carbon precursors, such as methane (CH₄) or acetylene (C₂H₂), under high temperatures (typically 900°C to 1000°C).
During the CVD process, the carbon atoms decompose and form a graphene sheet that grows layer by layer on the substrate. This results in high-purity graphene with superior electrical, mechanical, and thermal properties, which is ideal for a wide array of applications.
How Nanografi Uses CVD to Produce Graphene
Nanografi is a global leader in the production of high-quality graphene using CVD technology. The company uses state-of-the-art equipment to ensure precise control over the CVD process, producing graphene that is consistent in quality, size, and thickness. Here’s how Nanografi’s CVD graphene is produced:
- Substrate Preparation: The process begins with the selection of an appropriate metal substrate, such as copper or nickel, which provides a smooth surface for graphene growth. These metals are chosen because they provide excellent carbon atom deposition and ensure uniform graphene growth.
- Gas Precursor Introduction: The next step involves introducing a carbon precursor gas (typically methane or acetylene) into the reactor. Hydrogen or argon is often used to control the reaction, as they help regulate the process and reduce unwanted reactions.
- High-Temperature Reaction: Under controlled high temperatures (typically around 900°C-1000°C), the carbon precursor molecules decompose, and carbon atoms are deposited onto the metal substrate. Over time, these atoms self-assemble into graphene layers, forming a high-quality graphene sheet.
- Graphene Transfer: After the desired number of graphene layers has been grown, the graphene is transferred to a target substrate (such as silicon or polymer) through a process called wet transfer. This involves dissolving the copper substrate in a copper etching solution, leaving behind the graphene sheet.
- Quality Control: Nanografi conducts rigorous testing to ensure that the graphene produced through CVD meets high standards. They assess the graphene’s crystallinity, sheet size, defect density, and electrical properties to ensure the highest level of quality.
Properties of Nanografi’s CVD Graphene
Nanografi’s CVD graphene offers several remarkable properties that set it apart from other graphene production methods. These properties include:
1. High Electrical Conductivity
Graphene is renowned for its exceptional electrical conductivity, and Nanografi’s CVD graphene maintains this characteristic. The high-purity graphene produced through CVD ensures minimal defect density, allowing for efficient electron flow, which is crucial for applications like electronics, energy storage devices, and sensors.
2. Large Area and Uniformity
One of the challenges in graphene production is ensuring uniformity across large areas. Nanografi’s CVD method produces high-quality, large-area graphene sheets, which is essential for mass production applications such as flexible electronics and transparent conductive films.
3. High Mechanical Strength
CVD graphene is known for its incredible mechanical properties. With its tensile strength far surpassing that of steel, Nanografi’s CVD graphene can be used in applications that require materials with extreme durability and flexibility, such as wearable electronics and composites.
4. Thermal Conductivity
Graphene’s thermal conductivity is another outstanding feature. Nanografi’s graphene, produced via CVD, exhibits excellent heat dissipation properties, which is vital for high-performance electronics and energy storage systems, ensuring that devices remain cool during high-energy operations.
5. Customization and Doping Potential
Nanografi offers the potential for doping CVD graphene with various materials (e.g., nitrogen, boron, or oxygen) to tailor the material’s properties for specific applications. For example, doping with nitrogen can improve the electrocatalytic activity, making it suitable for fuel cells and batteries.
Applications of Nanografi’s CVD Graphene
The unique properties of Nanografi’s CVD graphene make it suitable for a wide range of applications across multiple industries. Here are some key areas where CVD graphene can be employed:
1. Energy Storage and Batteries
In supercapacitors and lithium-ion batteries, graphene is used to enhance charge capacity, charge-discharge efficiency, and cycle stability. Nanografi’s high-quality CVD graphene helps in enhancing battery performance, offering higher energy densities and faster charging times. It also plays a significant role in solid-state batteries by providing better conductivity and stability.
2. Flexible Electronics
Graphene’s flexibility, lightweight nature, and electrical conductivity make it ideal for flexible electronic devices, such as wearable tech, flexible displays, and solar cells. Nanografi’s CVD graphene, with its large-area production, is perfect for large-scale manufacturing of bendable circuits and transparent electrodes.
3. Sensors and Biosensors
Graphene’s high surface area and reactive edges make it an excellent material for sensors, particularly biosensors used in medical diagnostics. Nanografi’s graphene is often used in gas sensors, biosensors, and chemical sensors, where its properties allow for rapid detection of gases, diseases, or toxins.
4. Composites and Coatings
Graphene can significantly enhance the mechanical properties of composite materials. Nanografi’s CVD graphene is used in creating lightweight, strong, and conductive composites for aerospace, automotive, and construction applications. It’s also used in coatings to improve corrosion resistance, wear resistance, and thermal conductivity.
5. Water Filtration and Desalination
Nanografi’s CVD graphene has been explored for use in water filtration and desalination technologies, where its high surface area and ability to form ultra-thin membranes enable the removal of ions, molecules, and even salts from water. Graphene oxide membranes can efficiently filter nanoparticles and contaminants, providing access to cleaner water.
Challenges and Future Directions
While CVD graphene has remarkable potential, there are still challenges in scaling up production, reducing costs, and ensuring uniformity over large areas. Nanografi continues to focus on improving production techniques to make graphene more cost-effective for widespread use.
Future advancements in CVD graphene production may also include the development of 3D graphene structures, graphene-based devices, and more environmentally sustainable manufacturing methods.
Conclusion
Nanografi’s CVD graphene represents a major advancement in the production of high-quality graphene, thanks to its precise synthesis and customizable properties. With applications across a wide range of industries—electronics, energy storage, sensors, composites, and water filtration—CVD graphene is set to play a key role in the future of nanotechnology.
As Nanografi continues to push the boundaries of graphene production, the potential of this remarkable material will only expand, leading to even more innovative and sustainable solutions in the coming years.
