Graphene-Based Materials for Wastewater Treatment: Revolutionizing Water Purification
Water contamination is one of the biggest challenges faced by modern societies, especially with increasing industrialization, urbanization, and population growth. Efficient wastewater treatment has become essential not only to ensure the availability of clean water but also to protect the environment from harmful pollutants. Traditional methods of wastewater treatment, such as chemical coagulation, filtration, and membrane processes, often have limitations in terms of efficiency, cost, and environmental impact.
In recent years, graphene-based materials have emerged as promising solutions for addressing the limitations of conventional wastewater treatment technologies. Graphene, a single layer of carbon atoms arranged in a hexagonal lattice, is known for its exceptional properties, including high surface area, mechanical strength, conductivity, and chemical stability. When functionalized or combined with other materials, graphene-based materials demonstrate superior performance in the removal of pollutants from wastewater.
This article will explore the potential of graphene-based materials for wastewater treatment, including their mechanisms, benefits, challenges, and future applications.
What Are Graphene-Based Materials?
Graphene-based materials refer to various materials derived from or incorporating graphene in some form. These materials can include:
- Graphene Oxide (GO): Graphene oxide is a derivative of graphene that contains oxygen-containing groups such as epoxides, hydroxyls, and carboxyls. GO is hydrophilic, which makes it easier to disperse in water and modify for various applications.
- Reduced Graphene Oxide (rGO): When graphene oxide is reduced, the oxygen groups are removed, making the material more hydrophobic and conductive, which enhances its adsorptive properties.
- Graphene Composites: Graphene is often combined with other materials like carbon nanotubes (CNTs), metal oxides, and polymers to form composites with enhanced properties for specific wastewater treatment applications.
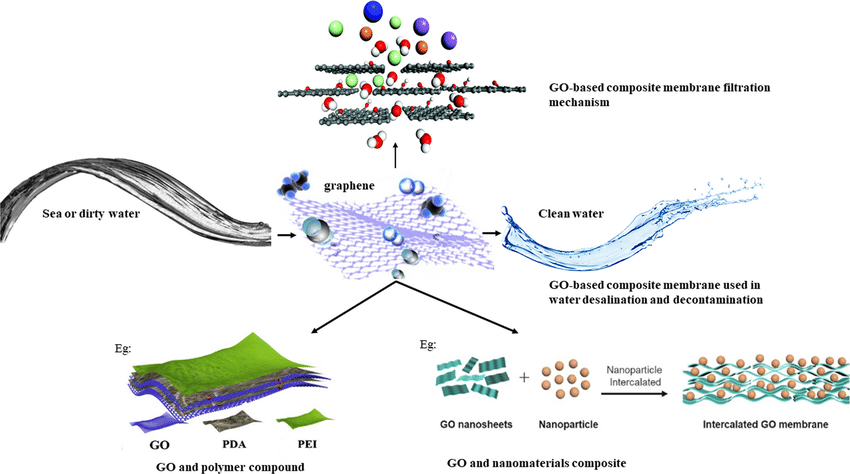
- Graphene-based Membranes: These are thin layers of graphene or graphene oxide used in filtration processes. They can selectively filter out contaminants based on size, charge, or chemical interactions.
- Graphene Aerogels: These lightweight, porous materials are made by incorporating graphene into a 3D network, offering high surface area and low density, which makes them ideal for adsorbing pollutants in water.
Mechanisms of Graphene-Based Materials in Wastewater Treatment
Graphene-based materials can tackle wastewater treatment in several ways, depending on their structure and functionalization. Here are some of the key mechanisms involved:
1. Adsorption
- Adsorption is the process by which pollutants (such as heavy metals, organic compounds, dyes, and pharmaceuticals) adhere to the surface of a material. Graphene-based materials, especially graphene oxide and reduced graphene oxide, exhibit high surface areas and functional groups that can effectively adsorb pollutants.
- For example, graphene oxide has abundant hydroxyl and carboxyl groups that can interact with metal ions, organic molecules, and dyes. This makes graphene oxide effective for removing contaminants like lead (Pb), cadmium (Cd), arsenic (As), and oil.
2. Filtration
- Graphene-based membranes are highly efficient at filtering out contaminants from water. The nano-porous structure of graphene allows for the filtration of small particles, including nanoparticles, bacteria, and viruses. These membranes can act as a physical barrier to larger contaminants while allowing water molecules to pass through.
- Additionally, graphene oxide membranes have shown the potential to selectively filter out certain contaminants based on their size or charge, a feature that can be fine-tuned to suit specific applications in wastewater treatment.
3. Catalysis
- Graphene-based catalysts have been developed to accelerate chemical reactions that break down organic pollutants. For example, graphene-based photocatalysts can be used to degrade organic pollutants under light irradiation. The high surface area and unique electronic properties of graphene enhance the efficiency of these catalytic processes.
- Graphene-metal oxide composites (e.g., graphene-TiO2) are also widely used for advanced oxidation processes (AOPs), which generate hydroxyl radicals to degrade toxic substances in wastewater.
4. Electrochemical Processes
- Graphene-based electrodes are increasingly used in electrochemical processes like electrocoagulation, electroflotation, and electrochemical oxidation. These processes rely on electrical currents to remove contaminants from water.
- Graphene’s high conductivity and large surface area make it an excellent material for electrode fabrication, enabling efficient removal of pollutants such as heavy metals and organic pollutants.
Benefits of Graphene-Based Materials for Wastewater Treatment
The integration of graphene-based materials into wastewater treatment technologies offers several advantages:
- High Surface Area and Porosity
- Graphene and its derivatives have an extremely high surface area, typically around 2630 m²/g for a single graphene sheet. This high surface area allows for the efficient adsorption of large quantities of pollutants, making graphene-based materials highly effective in wastewater treatment.
- Excellent Adsorption Capacity
- Due to their large surface area and chemical functionalities, graphene oxide and reduced graphene oxide can adsorb a variety of contaminants, including heavy metals, dyes, pharmaceuticals, and organic pollutants, effectively reducing water contamination.
- Cost-Effectiveness
- Graphene-based materials can be synthesized in a cost-effective manner, particularly with advances in scalable production techniques. For example, graphene oxide can be produced by oxidizing natural graphite, which is abundant and relatively inexpensive.
- The high efficiency of graphene-based materials means that less material is required to treat large volumes of wastewater, potentially lowering costs compared to conventional treatments.
- Versatility
- Graphene can be modified and functionalized to suit a variety of wastewater contaminants. The material can be easily adapted for specific applications such as removing heavy metals, oil spills, or organic pollutants. Additionally, the ability to combine graphene with other materials (like metals, polymers, or carbon nanotubes) enhances its performance for specific types of wastewater treatment.
- Reusability
- Graphene-based materials are highly durable and can be reused multiple times without significant degradation in performance. After adsorption, graphene-based materials can often be regenerated through simple washing or desorption techniques, making them suitable for continuous wastewater treatment processes.
- Energy-Efficiency
- Many graphene-based wastewater treatment technologies, such as membrane filtration and electrochemical processes, are more energy-efficient than traditional methods. This reduces the overall operational costs and environmental footprint of wastewater treatment plants.
Applications of Graphene-Based Materials in Wastewater Treatment
Graphene-based materials are being explored in a wide range of applications for improving wastewater treatment processes. Some key applications include:
1. Heavy Metal Removal
- Graphene oxide and reduced graphene oxide are particularly effective in adsorbing heavy metals such as lead (Pb), cadmium (Cd), arsenic (As), and chromium (Cr) from industrial wastewater. These metals can be toxic to human health and the environment, and their removal is a critical task in wastewater treatment.
2. Organic Pollutant Degradation
- Graphene-based materials, particularly when combined with metal oxides like titanium dioxide (TiO2), are used in advanced oxidation processes for the degradation of organic pollutants, including dyes, pharmaceuticals, and pesticides. These materials can break down complex organic compounds into less harmful byproducts.
3. Oil and Petroleum Waste Treatment
- Graphene aerogels and graphene composites are being used for the absorption of oils and petroleum-based pollutants. Their high porosity and oil-loving properties make them ideal for cleaning up oil spills and treating wastewater contaminated with hydrocarbons.
4. Water Desalination and Filtration
- Graphene oxide membranes are also being studied for desalination and water filtration. The high selectivity of graphene-based membranes allows them to filter out contaminants from water while allowing the passage of clean water molecules, making them ideal for seawater desalination and the treatment of wastewater with high salinity.
5. Electrochemical Water Treatment
- Graphene-based electrodes are employed in electrochemical water treatment techniques, such as electrocoagulation and electrochemical oxidation, for the removal of toxic metals, organic pollutants, and bacteria. The high conductivity of graphene enables faster and more efficient electrochemical reactions, improving the overall performance of the process.
Challenges and Future Directions
While the potential of graphene-based materials for wastewater treatment is immense, there are several challenges to overcome:
- Scalability: Producing graphene-based materials in large quantities with consistent quality and performance remains a challenge. As production methods improve, the cost of graphene will likely decrease, making it more accessible for industrial applications.
- Cost: Although graphene materials are becoming more cost-effective, the synthesis and functionalization processes can still be expensive compared to traditional wastewater treatment technologies.
- Environmental Impact: While graphene is non-toxic in many forms, there are concerns about the environmental impact of graphene-based materials when they are released into the environment, particularly at large scales. Further research is needed to ensure that graphene materials do not pose a risk to ecosystems.
Conclusion
Graphene-based materials represent a promising solution for improving wastewater treatment technologies. Their unique combination of high surface area, tunable properties, and exceptional performance in adsorbing pollutants makes them ideal candidates for removing contaminants such as heavy metals, organic pollutants, and oils from wastewater. As production methods advance and research into environmental impacts continues, graphene-based materials are likely to play a key role in the development of more efficient, sustainable, and cost-effective wastewater treatment solutions.
With ongoing advancements in nanotechnology and material science, graphene-based materials have the potential to transform the way we treat wastewater, ensuring cleaner water for future generations.
