Graphene Balls and Lithium-Ion Batteries: A New Era of Energy Storage
Graphene, a single layer of carbon atoms arranged in a two-dimensional honeycomb structure, has emerged as one of the most exciting materials in modern science and technology. Known for its exceptional electrical, thermal, and mechanical properties, graphene is now being explored for a wide range of applications, including energy storage. One of the most promising uses of graphene is in the development of lithium-ion (Li-ion) batteries, particularly through the innovative design of graphene balls. These structures have the potential to significantly improve the performance of conventional lithium-ion batteries, enhancing energy density, charging speed, and overall battery life.
This article delves into the role of graphene balls in lithium-ion batteries, exploring their properties, how they improve battery performance, and the potential for their future use in next-generation energy storage technologies.
What are Graphene Balls?
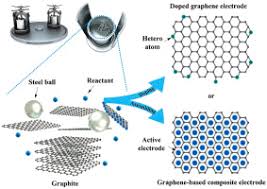
Graphene balls, also known as graphene-based microspheres, are spherical structures made from graphene oxide or reduced graphene oxide that are engineered at the microscale or nanoscale. These balls are typically formed by self-assembly processes, where graphene sheets spontaneously aggregate into three-dimensional spheres with highly porous interiors. The spheres can vary in size, but they are often in the range of 100 nm to several microns in diameter.
These graphene balls combine the high surface area, conductivity, and mechanical strength of graphene with the structural integrity of a three-dimensional spherical shape. Their porous nature allows for greater material loading, making them ideal candidates for use in batteries and other energy storage devices.
How Graphene Balls Improve Lithium-Ion Battery Performance
The addition of graphene balls to lithium-ion batteries can significantly improve various key performance metrics, including energy density, charging rates, cycle stability, and thermal conductivity. Here’s how graphene balls contribute to these improvements:
1. Enhanced Energy Density
The high surface area of graphene, coupled with the porous structure of graphene balls, provides more surface area for the storage of lithium ions. This increased surface area allows for greater ion storage capacity and facilitates the formation of a larger charge/discharge cycle in the battery. As a result, graphene balls can boost the energy density of lithium-ion batteries, enabling them to store more energy in the same amount of space, which is critical for applications like electric vehicles (EVs) and portable electronics.
2. Faster Charging and Discharging Rates
One of the key advantages of graphene balls is their ability to accelerate ion transport. The high electrical conductivity of graphene allows for faster movement of electrons during the charge and discharge cycles. Additionally, the three-dimensional structure of graphene balls provides multiple pathways for the movement of lithium ions, reducing internal resistance and speeding up the charging and discharging processes. This leads to faster charging times, which is a crucial factor for consumer electronics and electric vehicles.
3. Improved Cycle Life
Lithium-ion batteries typically suffer from capacity degradation over time due to volume expansion and contraction during cycling. This can cause mechanical stress on the anode and cathode materials, leading to cracking and material loss. Graphene balls can help mitigate this issue because their flexible structure can accommodate expansion and contraction without compromising structural integrity. This flexibility helps to enhance the cycle life of batteries, allowing them to endure many more charge-discharge cycles compared to conventional batteries.
4. Thermal Conductivity
Graphene’s high thermal conductivity can also be beneficial for improving the thermal stability of lithium-ion batteries. During the charging and discharging cycles, batteries generate heat, and excessive heat can damage battery materials, reducing efficiency and lifespan. The incorporation of graphene balls helps to distribute and dissipate heat more efficiently, ensuring that the battery stays within an optimal temperature range. This improved heat management contributes to longer battery life and greater safety.
5. Increased Stability and Safety
Graphene balls can also improve the chemical stability and safety of lithium-ion batteries. The ability of graphene balls to create a stable, conductive network within the battery ensures that the material is less likely to degrade or experience short-circuiting, which can lead to battery fires or explosions. Additionally, the chemical properties of graphene enhance the overall stability of the battery, reducing the risk of thermal runaway.
Synthesis of Graphene Balls for Lithium-Ion Batteries
The production of graphene balls involves various techniques, each designed to control the size, morphology, and surface properties of the balls. Here are the common methods used for synthesizing graphene balls:
1. Chemical Vapor Deposition (CVD)
CVD is a well-known technique for producing high-quality graphene. In this method, a carbon-containing gas, such as methane or acetylene, is decomposed at high temperatures in the presence of a catalyst. The carbon atoms then deposit on a substrate, forming graphene nanosheets, which self-assemble into graphene balls. The advantage of CVD is that it can produce highly uniform and pure graphene balls, but the process can be expensive and requires specialized equipment.
2. Hydrothermal Synthesis
In this method, a precursor material such as graphene oxide is dissolved in water along with other chemicals, and the mixture is subjected to high pressure and temperature in a sealed container (a hydrothermal reactor). Under these conditions, the graphene oxide is reduced to graphene and forms self-assembled spheres. Hydrothermal synthesis is more cost-effective than CVD and can produce large quantities of graphene balls, though it may not always result in the highest quality material.
3. Solvothermal Synthesis
Similar to hydrothermal synthesis, solvothermal synthesis involves the reaction of graphene oxide with organic solvents, rather than water, under high temperature and pressure. This method allows for greater control over the size and morphology of the graphene balls and can be used to incorporate additional materials, such as polymers or metal nanoparticles, to further enhance the properties of the graphene balls for battery applications.
4. Self-Assembly
Self-assembly is a technique that exploits the natural tendency of graphene oxide or reduced graphene oxide sheets to organize into stable structures. Under controlled conditions, these sheets form microspherical aggregates. This method is particularly attractive because it is simple and scalable, and it avoids the need for harsh chemical treatments or expensive equipment.
Applications of Graphene Balls in Lithium-Ion Batteries
Graphene balls are increasingly being utilized to improve the performance of lithium-ion batteries in various applications, such as:
1. Electric Vehicles (EVs)
As the demand for electric vehicles grows, improving the energy storage capabilities of batteries becomes crucial. The use of graphene balls in EV batteries can enable faster charging, higher energy density, and longer battery life, making electric vehicles more convenient, cost-effective, and environmentally friendly.
2. Consumer Electronics
Smartphones, laptops, and wearable devices are constantly pushing the limits of battery performance. Graphene balls can enhance the performance of lithium-ion batteries in consumer electronics, providing longer-lasting power and faster charging times, all while maintaining a compact form factor.
3. Renewable Energy Storage
Graphene balls can also be used in energy storage systems, such as those that store energy from solar or wind power. Batteries that incorporate graphene balls can improve the efficiency and reliability of energy storage solutions, enabling better utilization of renewable energy sources.
4. Grid Storage and Large-Scale Energy Solutions
Large-scale grid storage applications require batteries that can store large amounts of energy and deliver reliable performance over time. Graphene balls in lithium-ion batteries can contribute to the development of more efficient grid storage systems by offering improved charge retention and faster charge/discharge cycles.
Future Outlook and Challenges
The potential of graphene balls to transform lithium-ion battery technology is vast, but several challenges remain. The scalability of production methods, the cost of materials, and the need for further optimization of performance metrics such as cycle life and safety must be addressed before graphene-based batteries can be widely commercialized.
That said, ongoing research and development in this area are showing great promise. As techniques for synthesizing and incorporating graphene balls into lithium-ion batteries continue to improve, we can expect to see significant advancements in energy storage devices that are lighter, more efficient, and long-lasting.
Conclusion
Graphene balls are a promising addition to the world of energy storage technologies, particularly for lithium-ion batteries. By enhancing key performance factors such as energy density, charging speed, cycle stability, and thermal conductivity, graphene balls are poised to play a crucial role in the development of the next generation of energy storage solutions. With further advancements in synthesis techniques and material optimization, graphene balls could soon become a standard feature in consumer electronics, electric vehicles, and renewable energy storage systems, ushering in a new era of more efficient, faster-charging, and longer-lasting batteries.
