Explained: Graphene, Graphene Oxide, and Reduced Graphene Oxide and Their Applications
Introduction
In the world of advanced materials, graphene, graphene oxide (GO), and reduced graphene oxide (rGO) have emerged as highly promising substances with a wide range of applications. These materials are distinct forms of graphene, a two-dimensional (2D) carbon nanomaterial that has garnered significant attention due to its unique properties. Graphene, first isolated in 2004, is now considered one of the most significant discoveries in materials science, as it has applications across multiple fields including electronics, energy, healthcare, and environmental sustainability.
In this article, we will explore the differences between graphene, graphene oxide, and reduced graphene oxide, as well as their unique applications in various industries.
1. What is Graphene?
Graphene is a single layer of carbon atoms arranged in a two-dimensional honeycomb lattice. This structure gives graphene remarkable properties:
- Electrical Conductivity: Graphene is an excellent conductor of electricity, making it ideal for use in electronic devices.
- Mechanical Strength: Despite being just one atom thick, graphene is incredibly strong—about 200 times stronger than steel by weight.
- Thermal Conductivity: It has high thermal conductivity, meaning it can efficiently transfer heat.
- Flexibility: Graphene is incredibly flexible, allowing it to bend and stretch without breaking.
- High Surface Area: A single sheet of graphene has a surface area of approximately 2630 m² per gram, making it ideal for applications like energy storage.
Applications of Graphene
- Electronics: Graphene is used in the development of flexible electronic devices, touchscreens, and transparent conductive films for display technologies.
- Energy Storage: Due to its high surface area, graphene is used in supercapacitors, batteries, and fuel cells to improve energy storage and charging capabilities.
- Composites: Graphene is incorporated into materials like carbon fiber composites, enhancing their strength and reducing weight.
- Biomedical: In medicine, graphene’s biocompatibility allows it to be used in drug delivery, biosensors, and tissue engineering.
2. What is Graphene Oxide (GO)?
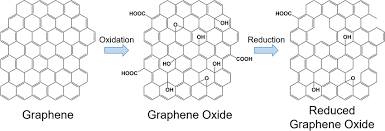
Graphene oxide (GO) is a derivative of graphene that has oxygen-containing functional groups (such as hydroxyl, epoxide, and carboxyl groups) attached to its surface. These functional groups make GO more hydrophilic (water-attracting), which makes it easier to disperse in solvents, unlike pure graphene, which is typically hydrophobic.
Properties of Graphene Oxide
- Surface Chemistry: The oxygenated groups on GO provide a high level of surface reactivity, enabling it to be further modified with various molecules and ions.
- Electrical Insulator: Unlike graphene, graphene oxide is typically an electrical insulator due to the disruption of the electron flow by the oxygenated groups.
- Solubility: GO is much easier to disperse in water and other solvents, which makes it a more versatile material for various applications.
Applications of Graphene Oxide
- Water Filtration: The hydrophilic nature of GO makes it an excellent candidate for water purification and desalination. GO membranes have shown great promise in filtering small molecules and ions due to their well-structured pores.
- Energy Storage: GO can be used to enhance the performance of supercapacitors and batteries by acting as a conductive scaffold and improving charge storage capacity.
- Sensors and Biosensors: GO is widely used in the development of chemical sensors, biosensors, and environmental monitoring tools due to its large surface area and ease of functionalization.
- Drug Delivery: GO’s ability to load various molecules makes it suitable for targeted drug delivery systems, where it can carry drugs or genes to specific parts of the body.
- Coatings and Paints: The incorporation of GO into coatings enhances their properties, making them more durable and corrosion-resistant.
3. What is Reduced Graphene Oxide (rGO)?
Reduced graphene oxide (rGO) is obtained by reducing graphene oxide, typically through chemical, thermal, or electrochemical reduction methods. This process removes some of the oxygen-containing groups on the GO sheet, restoring the electrical conductivity closer to that of pure graphene.
Properties of Reduced Graphene Oxide
- Restored Electrical Conductivity: rGO possesses improved electrical conductivity compared to GO, but it is not as conductive as pure graphene.
- Partial Oxygen Groups: While rGO retains some oxygenated groups, the reduction process leads to fewer oxygen-containing functional groups than in GO, making it more hydrophobic and conductive.
- Porosity: The reduction process also alters the surface structure, creating pores that are useful for energy storage applications.
Applications of Reduced Graphene Oxide
- Supercapacitors: rGO is used in supercapacitors for energy storage due to its high surface area and enhanced conductivity compared to GO.
- Flexible Electronics: rGO is integrated into flexible electronics because of its mechanical flexibility and conductivity.
- Sensors: rGO is used to enhance the sensitivity of electrochemical sensors for detecting gases, biological agents, and environmental pollutants.
- Water Purification: Similar to GO, rGO can be used for water filtration applications, but its reduced form offers better mechanical properties and recyclability.
- Composites: rGO is incorporated into composites to enhance mechanical strength and conductivity, useful for applications in aerospace, automotive, and construction industries.
4. Comparison of Graphene, Graphene Oxide, and Reduced Graphene Oxide
| Property | Graphene | Graphene Oxide (GO) | Reduced Graphene Oxide (rGO) |
|---|---|---|---|
| Electrical Conductivity | Very high | Low (insulator) | Moderate to high |
| Surface Area | Extremely high (2630 m²/g) | High | Moderate to high |
| Solubility | Insoluble in water | Soluble in water and solvents | Slightly soluble, hydrophobic |
| Mechanical Strength | Exceptional | Moderate | High |
| Functional Groups | None | Oxygenated functional groups | Reduced oxygen groups |
| Applications | Electronics, Energy, Composites | Water filtration, Sensors, Drug delivery | Supercapacitors, Flexible electronics, Sensors |
5. Applications of Graphene, Graphene Oxide, and Reduced Graphene Oxide
Graphene Applications
- Electronics: Used in transistors, high-speed circuits, OLEDs, and graphene-based sensors.
- Energy Storage: Graphene is a key material in next-generation supercapacitors, batteries, and fuel cells.
- Composites: Graphene enhances the strength and durability of composite materials used in aerospace, automotive industries, and sports equipment.
Graphene Oxide Applications
- Water Purification: GO-based membranes are highly effective in separating contaminants from water.
- Medical: GO is used for drug delivery and biosensing applications due to its large surface area and reactivity.
- Energy: GO is used in supercapacitors and batteries, as it enhances charge storage by providing a conductive network.
Reduced Graphene Oxide Applications
- Energy Storage: rGO’s improved conductivity makes it ideal for use in supercapacitors and batteries with enhanced power and energy densities.
- Flexible Electronics: rGO is widely used in flexible displays, wearable devices, and electronic skins.
- Sensors: rGO’s high surface area and conductivity make it ideal for electrochemical sensors used in environmental monitoring and healthcare.
Conclusion
Graphene, graphene oxide, and reduced graphene oxide are materials that offer distinct properties, making them suitable for a wide range of applications. While graphene excels in electrical conductivity and mechanical strength, graphene oxide provides unique functional properties, especially in water purification and biomedical applications. Reduced graphene oxide, on the other hand, balances conductivity and functionality, making it ideal for energy storage and sensor technologies. As research continues, these materials are expected to play a key role in advancing technologies across industries, from electronics to healthcare, sustainability, and beyond.
