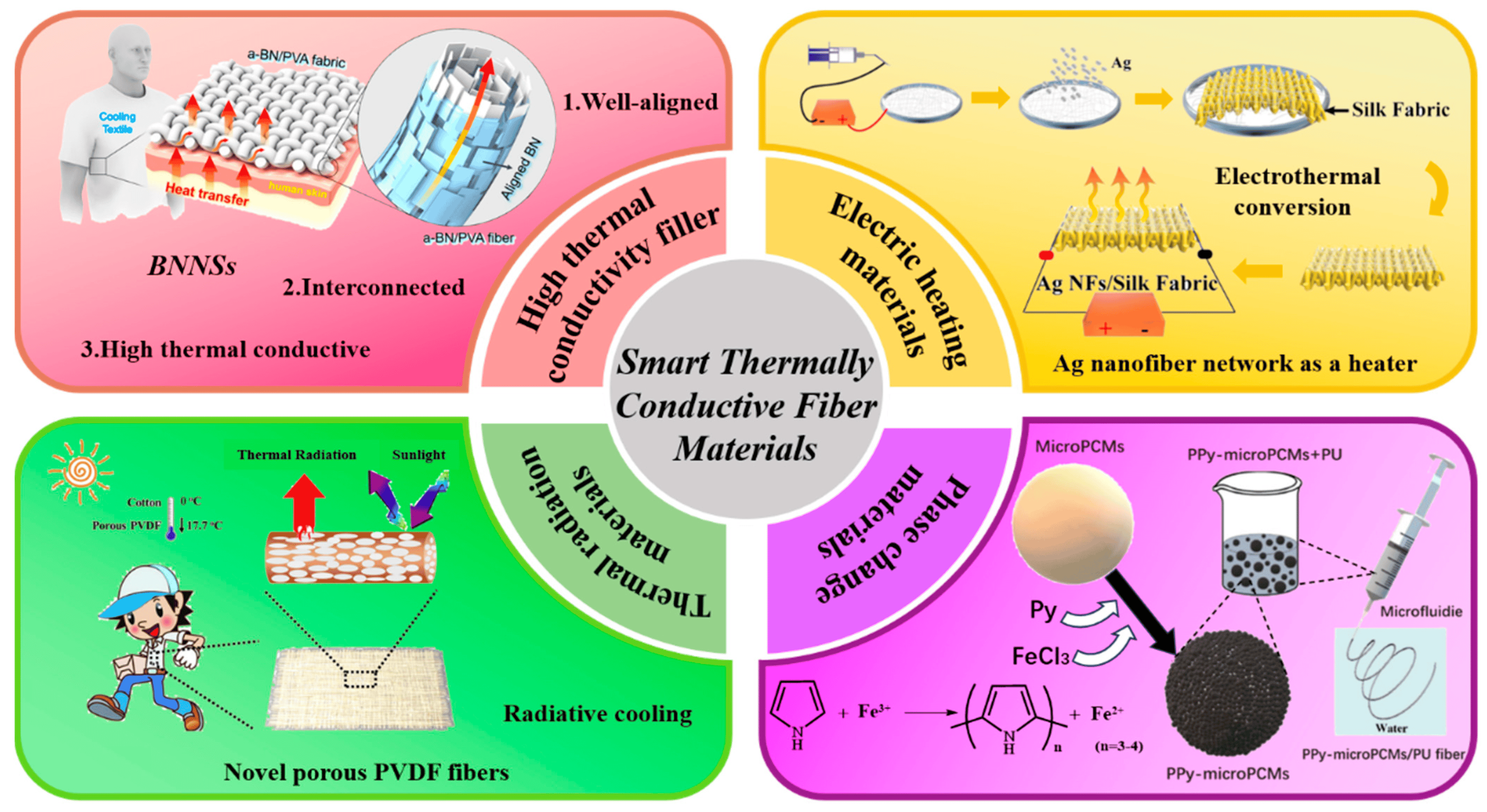
Conductive Materials and Their Industrial Applications
Conductive materials are substances that allow the flow of electric charge through them, playing a pivotal role in various industrial sectors. From electronics to energy transmission and biomedical devices, the demand for high-performance conductive materials has surged in recent years. These materials, which include metals, polymers, ceramics, and composites, possess exceptional electrical conductivity, making them essential in the development of advanced technologies and the optimization of industrial processes.
This article explores the various types of conductive materials, their properties, and the wide range of industrial applications in which they are used, emphasizing their importance in industries like electronics, energy, automotive, healthcare, and construction.
Types of Conductive Materials
Conductive materials are typically classified based on their composition and the level of conductivity they exhibit. Here are the main types:
1. Metals
- Copper (Cu): Copper is one of the most widely used conductive materials due to its high electrical conductivity, corrosion resistance, and relative abundance. It is commonly used in wiring, circuit boards, and power distribution systems.
- Aluminum (Al): While aluminum has slightly lower electrical conductivity compared to copper, it is still widely used in power cables, electric transmission lines, and lightweight electrical systems due to its low cost and lightweight nature.
- Gold (Au) and Silver (Ag): Both gold and silver are highly conductive metals, but they are more expensive. Gold is often used in high-precision electronic devices, such as connectors and semiconductor circuits, while silver is used in high-performance electrical contacts.
2. Polymers
- Conductive Polymers: Polymers are generally insulative materials, but conductive polymers are synthesized to carry electricity. These materials, such as polypyrrole, polyaniline, and PEDOT:PSS, are lightweight, flexible, and corrosion-resistant. Conductive polymers are widely used in applications like flexible electronics, antistatic coatings, and sensor technologies.
- Carbon-based Conductive Polymers: These polymers are often combined with carbon nanotubes or graphene to improve their conductivity. They are particularly useful in wearable electronics and printed circuit boards.
3. Ceramics
- Conductive Ceramics: These are materials that have been engineered to exhibit electrical conductivity, unlike traditional ceramics. Some of the most notable conductive ceramics include tungsten carbide (WC), manganese dioxide (MnO₂), and yttrium-stabilized zirconia (YSZ). These materials are used in sensors, capacitors, and high-temperature applications.
- Superconducting Ceramics: Certain ceramics, like YBCO (Yttrium Barium Copper Oxide), exhibit superconductivity at relatively high temperatures and are used in magnetic resonance imaging (MRI), maglev trains, and power transmission systems.
4. Composites
- Conductive Composites: Composites made from a combination of conductive materials, such as carbon fibers, carbon nanotubes, and graphene, embedded within a non-conductive matrix, have emerged as highly effective materials. These composites offer superior mechanical strength combined with electrical conductivity and are used in various sectors such as aerospace, automotive, and construction.
Properties of Conductive Materials
The primary properties that define the performance of conductive materials include:
- Electrical Conductivity: The ability of a material to allow electric charge to flow. It is typically measured in siemens per meter (S/m). Materials with high conductivity, such as copper, are used for transmitting electrical signals and energy over long distances.
- Thermal Conductivity: Many conductive materials also exhibit high thermal conductivity, meaning they can efficiently transfer heat. For example, metals like copper and aluminum are commonly used in heat sinks and cooling systems in electronics.
- Mechanical Strength: The ability to withstand physical stress without breaking or deforming. This is especially important for materials used in structural applications or in devices that undergo repeated mechanical stress, such as automotive components.
- Corrosion Resistance: The resistance to chemical degradation due to environmental exposure. Materials such as gold and silver have excellent corrosion resistance, making them ideal for sensitive electronics and connectors.
- Flexibility: Conductive materials like conductive polymers and composites provide flexibility, making them suitable for applications in wearable electronics and smart textiles.
Industrial Applications of Conductive Materials
1. Electronics and Semiconductors
The electronics industry is perhaps the largest consumer of conductive materials. Some common applications include:
- Printed Circuit Boards (PCBs): Conductive materials like copper and silver are used in PCBs for connecting electrical components. Conductive polymers and carbon nanotubes are also used in flexible and printed electronics.
- Transistors and Microchips: Conductive materials are used in the fabrication of transistors and microchips that are at the core of modern computing devices like smartphones, laptops, and tablets.
- Batteries and Energy Storage: Conductive materials, especially carbon-based materials and lithium compounds, are crucial in batteries, supercapacitors, and energy storage systems. They are used to enhance the charge storage capacity and efficiency of energy storage devices.
2. Energy and Power Systems
- Power Transmission Lines: Copper and aluminum are widely used for power cables and transmission lines due to their high electrical conductivity, low resistance, and ability to carry high amounts of electrical energy over long distances.
- Solar Panels: Conductive materials, such as silver and copper, are used in the construction of photovoltaic cells to conduct the electricity generated by solar panels.
- Fuel Cells: Conductive ceramics and graphene-based materials are being utilized in the development of fuel cells, where they help in the conversion of chemical energy into electrical energy.
3. Automotive and Aerospace
- Electric Vehicles (EVs): Conductive materials are crucial in the development of electric vehicle batteries and powertrains, including the use of graphene composites for lighter, more efficient components.
- Sensors: Conductive polymers and carbon nanotubes are increasingly being used in sensors for monitoring various parameters like temperature, pressure, and gas composition in vehicles and aircraft.
- Antistatic Coatings: In automotive coatings and aerospace applications, conductive coatings prevent the accumulation of static electricity, reducing the risk of sparks and improving the performance of electronic components.
4. Healthcare and Biomedical Applications
- Biosensors: Conductive materials, particularly conductive polymers and carbon nanotubes, are used in the development of biosensors for detecting biomolecules such as glucose, oxygen levels, and enzymes for diagnostics and monitoring.
- Electrocardiogram (ECG) and Electroencephalogram (EEG) Electrodes: Gold and silver are used in electrodes for ECG and EEG to monitor heart and brain activity due to their excellent conductivity and biocompatibility.
- Implantable Devices: Conductive materials are used in the development of implantable medical devices such as pacemakers and neurostimulation devices, where electrical conduction is essential for their function.
5. Construction and Infrastructure
- Conductive Coatings: In the construction industry, conductive materials like graphene and carbon nanotubes are used in coatings for antistatic flooring, heating systems, and smart windows. These materials can help regulate indoor temperature and even convert solar energy into electricity.
- Smart Infrastructure: Conductive materials are essential in the development of smart roads and buildings. Conductive concrete can be used to charge electric vehicles or heat roads in winter to prevent ice formation.
6. Textiles and Wearable Technology
- Smart Fabrics: Conductive materials are used in wearable electronics and smart textiles, such as fitness trackers, health monitoring systems, and smart clothing. Conductive threads and conductive fabrics enable the integration of sensors, batteries, and circuitry into clothing.
Challenges and Future Trends
While conductive materials have revolutionized several industries, there are challenges that need to be addressed for continued advancement:
- Cost and Scalability: High-performance conductive materials, especially those based on graphene and carbon nanotubes, can be expensive to produce at scale. Reducing costs and improving production techniques are key to making these materials more accessible.
- Durability and Reliability: Many conductive materials, especially those used in flexible electronics and wearables, must be designed to withstand repeated use and environmental stress without degrading.
- Environmental Impact: Some conductive materials, such as silver and gold, are costly and less sustainable. Researchers are exploring alternatives like carbon-based materials and biodegradable conductive polymers to reduce the environmental footprint.
- Integration with Other Materials: As industries demand more multifunctional materials, researchers are working to develop composite materials that combine high conductivity with other desirable properties, such as strength, flexibility, and biocompatibility.
Conclusion
Conductive materials are the backbone of modern technology, enabling the operation of everything from smart devices to renewable energy systems. Their applications span across numerous industries, including electronics, energy, automotive, healthcare, and construction. With continued advancements in material science, innovative conductive materials like graphene, conductive polymers, and carbon composites are set to drive the future of technologies that require superior electrical conductivity, flexibility, and strength. The challenges of cost, scalability, and environmental impact are being actively addressed, paving the way for more efficient, sustainable, and advanced conductive solutions in the future.
