Battery Cooling with Nanotechnology: Enhancing Efficiency and Lifespan
The rapid development of energy storage technologies has made batteries an essential component in numerous applications, from electric vehicles (EVs) to consumer electronics. One of the most significant challenges associated with batteries is heat generation during charging and discharging cycles. Excessive heat not only reduces battery efficiency but can also lead to faster degradation, reduced lifespan, and safety risks such as overheating or thermal runaway.
To address these challenges, nanotechnology has emerged as a promising solution for improving battery cooling systems. By leveraging the unique properties of nanomaterials, scientists and engineers can enhance the performance, thermal management, and safety of modern batteries, especially as demand for high-capacity, fast-charging, and long-lasting batteries continues to grow.
In this article, we explore the role of nanotechnology in battery cooling, how it works, the types of nanomaterials being used, and the future potential of these technologies.
Why Battery Cooling is Crucial
Heat is generated in batteries due to electrochemical reactions that occur during charging and discharging cycles. This heat, if not properly dissipated, can lead to several issues:
- Efficiency Loss:
- High temperatures can increase internal resistance, which results in energy losses. This reduces the overall efficiency of the battery.
- Reduced Battery Lifespan:
- Excessive heat accelerates the degradation of key battery components, such as the electrodes and electrolyte, which leads to a shortened lifespan of the battery.
- Safety Concerns:
- In extreme cases, overheating can result in thermal runaway, a condition in which the battery becomes uncontrollably hot and may catch fire or explode.
For these reasons, an effective cooling system is critical to the long-term performance and safety of batteries. Traditional methods of battery cooling, such as air cooling or liquid cooling, have limitations in terms of energy efficiency and space constraints, especially in high-performance applications. Nanotechnology, with its ability to manipulate materials at the atomic and molecular scale, offers a promising solution for overcoming these challenges.
How Nanotechnology Enhances Battery Cooling
Nanotechnology can improve battery cooling in a variety of ways, primarily through the use of nanomaterials that possess unique thermal and mechanical properties. Here are the key mechanisms by which nanotechnology is enhancing battery cooling systems:
1. Improved Thermal Conductivity with Nanomaterials
One of the key advantages of nanomaterials is their exceptionally high thermal conductivity. This property allows them to efficiently transfer heat away from the battery cells and distribute it more evenly. Several nanomaterials are being explored for use in thermal management systems in batteries:
- Carbon Nanotubes (CNTs):
- Carbon nanotubes have outstanding thermal conductivity and can be integrated into battery electrodes, current collectors, and cooling components to enhance heat dissipation. CNTs can direct heat flow away from the core of the battery, preventing overheating.
- Graphene:
- Like CNTs, graphene also exhibits remarkable thermal conductivity. Graphene-based composites are increasingly used in battery cooling solutions due to their ability to conduct heat efficiently while being lightweight and flexible. Graphene is being studied for applications in both thermal management films and conductive foams.
- Metal Nanoparticles:
- Nanoparticles of metals such as copper and silver have excellent thermal conductivity and can be incorporated into battery cooling systems. Metal nanoparticle-based heat sinks are being developed to help dissipate heat away from battery cells, improving efficiency and safety.
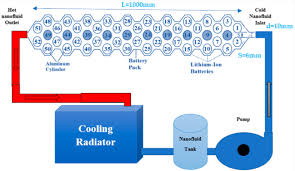
2. Nanofluids for Enhanced Cooling Efficiency
Nanofluids are a class of liquid suspensions that contain nanoparticles, which significantly improve the thermal properties of the fluid. These fluids can be circulated through battery cooling systems to enhance heat removal.
- Nanofluids containing carbon nanotubes (CNTs), graphene oxide (GO), or metal oxide nanoparticles have been shown to improve heat transfer rates significantly compared to conventional coolants. These nanofluids can be used in liquid cooling systems to absorb and carry heat away from the battery cells, ensuring that the temperature remains within safe limits.
- Advantages:
- Higher heat transfer efficiency: Nanofluids have an enhanced ability to absorb and dissipate heat.
- Compact cooling systems: Due to the enhanced thermal properties, smaller amounts of nanofluid can be used, resulting in more compact cooling designs.
- Longer battery life: By maintaining an optimal temperature, nanofluids reduce the risk of overheating, thereby prolonging the life of the battery.
3. Phase Change Materials (PCMs) with Nanotechnology
Phase change materials (PCMs) are substances that absorb or release latent heat as they change from solid to liquid or vice versa. These materials can be used to absorb the excess heat generated by batteries during charging and discharging cycles. By incorporating nanotechnology into the design of PCMs, the heat-absorbing capabilities of these materials can be improved.
- Nanostructured PCMs: The incorporation of nanoparticles into PCMs increases the thermal conductivity and heat storage capacity, making them more efficient at absorbing heat during periods of high battery activity.
- Thermal regulation: When the temperature of the battery rises above a certain threshold, PCMs undergo a phase change, storing the excess heat and preventing the battery from overheating. When the temperature drops, the PCM releases the stored heat, helping to maintain a stable operating temperature for the battery.
4. Advanced Battery Cooling Coatings
Nanocoatings are thin layers of nanomaterials that can be applied to battery components to enhance thermal regulation. These coatings can be used on battery electrodes, casings, and conductive materials to improve heat resistance and heat dissipation.
- Thermal management coatings: Nanocomposite coatings that include nano-sized ceramic particles or graphene are being developed to prevent heat build-up and reduce temperature fluctuations within the battery. These coatings are highly effective at reflecting heat and distributing it evenly across the surface, preventing local hotspots that can damage the battery.
- Durability: Nanocoatings not only help in thermal management but also improve the durability and longevity of the battery by protecting it from external environmental factors, such as moisture and corrosion, which can affect performance.
Applications of Nanotechnology in Battery Cooling
The use of nanotechnology for battery cooling is particularly important in the following high-performance industries:
1. Electric Vehicles (EVs)
As the adoption of electric vehicles continues to grow, the need for efficient and reliable battery systems becomes more crucial. In EVs, batteries are subjected to frequent charging and discharging cycles, often under high loads, which generates significant amounts of heat. Efficient battery cooling systems using nanomaterials help maintain optimal operating temperatures, ensuring faster charging, longer driving ranges, and safer operation.
2. Consumer Electronics
In smartphones, laptops, and wearables, batteries are essential for portability and performance. Nanotechnology-based cooling solutions can prevent overheating, extend battery life, and improve overall device performance by reducing heat build-up in these compact devices.
3. Energy Storage Systems
Large-scale energy storage systems (such as those used in solar power plants or grid storage) often require efficient thermal management to prevent overheating during high-power charging and discharging cycles. Nanotechnology-enabled cooling systems, including nanofluids and graphene-based coatings, are being integrated into these systems to ensure their longevity and stability.
Challenges and Future Outlook
While nanotechnology offers exciting opportunities for battery cooling, there are still challenges to overcome:
- Cost: The production of advanced nanomaterials can be expensive, and scaling up these technologies for mass-market applications requires significant investment.
- Long-term stability: The long-term performance and stability of nanomaterials under real-world conditions (e.g., high temperatures, mechanical stress) need to be thoroughly tested.
- Regulatory concerns: As with all nanotechnologies, there are concerns regarding the environmental impact and safety of using nanoparticles in large-scale applications.
However, as research progresses and production techniques improve, the role of nanotechnology in battery cooling is expected to expand significantly, resulting in smaller, lighter, more efficient, and longer-lasting batteries.
Conclusion
Nanotechnology holds the key to addressing some of the most pressing challenges in battery cooling by improving thermal conductivity, heat dissipation, and overall safety. As the demand for electric vehicles, renewable energy storage, and portable electronics grows, nanomaterials will play an increasingly critical role in ensuring that batteries remain efficient, durable, and safe.
By leveraging the unique properties of nanofluids, nanocoatings, and advanced thermal management systems, nanotechnology promises to revolutionize the way we manage battery heat, helping to unlock the full potential of next-generation energy storage technologies.
