Applications of Graphene in Metal-Air Batteries
Introduction
Metal-air batteries, including lithium-air, zinc-air, and sodium-air batteries, have emerged as promising candidates for next-generation energy storage technologies due to their high energy densities and potential for low-cost, long-lasting performance. However, challenges like poor conductivity, limited lifespan, and low efficiency need to be addressed to realize their full potential. This is where graphene, a material renowned for its exceptional electrical conductivity, mechanical strength, and surface area, comes into play.
In this article, we explore the various applications of graphene in metal-air batteries, focusing on how it is helping to overcome the inherent limitations of these batteries, enhancing performance, and contributing to the development of next-generation energy storage solutions.
What is Graphene?
Graphene is a single layer of carbon atoms arranged in a two-dimensional hexagonal lattice. It is known for its remarkable properties:
- Electrical conductivity: Graphene exhibits excellent electrical conductivity, making it ideal for improving the performance of various types of batteries.
- Mechanical strength: Despite being extremely thin, graphene is one of the strongest known materials.
- High surface area: Graphene has a very high surface area (~2630 m²/g), which is beneficial for charge storage.
- Flexibility: Graphene’s flexibility makes it suitable for flexible electronics and batteries.
Because of these properties, graphene has found applications in multiple fields, including energy storage, electronics, and environmental sustainability.
Graphene in Metal-Air Batteries: The Key Advantages
In metal-air batteries, the electrolyte and metal electrodes are exposed to the air (oxygen from the atmosphere), which allows these batteries to achieve much higher energy densities compared to traditional lithium-ion batteries. However, some significant challenges still persist:
- Low conductivity of the air cathode.
- Limited efficiency due to poor electron and ion transfer.
- Instability of the metal anodes over long usage cycles.
- Corrosion issues at the air cathode.
Graphene addresses these challenges in several critical ways:
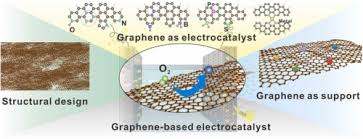
1. Enhancing Conductivity of the Air Cathode
In metal-air batteries, the air cathode plays a pivotal role in the electrochemical reaction by facilitating the reduction of oxygen to form hydroxide or peroxide ions. However, air cathodes often suffer from low conductivity, which can reduce the efficiency of the battery.
- Graphene as Conductive Additive: By integrating graphene into the air cathode, researchers have significantly improved its electrical conductivity. Graphene’s high electron mobility helps facilitate the transfer of electrons during the reduction and oxidation reactions at the air cathode, thereby improving the overall performance of metal-air batteries.
- Graphene-Carbon Composite Cathodes: Graphene is often combined with carbon-based materials, such as carbon nanotubes or graphene oxide, to further enhance conductivity. This composite material can provide both mechanical strength and electrical conductivity, allowing for more efficient electron transfer during the discharge process.
2. Improving the Stability of the Metal Anode
The metal anode (e.g., zinc in zinc-air batteries or lithium in lithium-air batteries) undergoes significant challenges during cycling, such as dendrite growth and corrosion, which leads to performance degradation and short battery life. Graphene can be used to address these issues in the following ways:
- Graphene-Coated Metal Anodes: One common approach is to coat metal anodes with a layer of graphene. This layer prevents the formation of dendrites, which can short-circuit the battery. Graphene’s smooth and stable structure provides a uniform interface that minimizes metal electrode degradation.
- Enhanced Ion and Electron Transport: Graphene enhances ion and electron transport in metal anodes, reducing the internal resistance of the battery and helping to maintain a more consistent charge-discharge cycle. This leads to a more stable and efficient battery performance over time.
3. Increasing Energy Density
Energy density is a critical factor in determining the effectiveness of metal-air batteries, especially when compared to lithium-ion batteries. The high surface area and conductive properties of graphene make it an ideal material for enhancing the energy density of metal-air batteries.
- Graphene-Based Electrode Materials: The incorporation of graphene into the electrodes of metal-air batteries increases the active surface area, which enables more electrochemical reactions to occur within the same volume. This results in a higher specific capacity and improved overall energy density.
- Enhanced Oxygen Reduction Reaction (ORR): In metal-air batteries, the oxygen reduction reaction (ORR) at the air cathode is critical to battery performance. Graphene, especially when functionalized or doped with other materials, enhances the ORR, increasing the efficiency and energy density of metal-air batteries.
4. Enhancing Cycle Life
One of the major disadvantages of metal-air batteries is their limited cycle life, primarily due to the degradation of the air cathode and metal anodes during repeated charge-discharge cycles. Graphene can significantly enhance the cycle life of metal-air batteries by improving the structural integrity and stability of key components:
- Graphene as a Structural Reinforcement: Graphene provides structural reinforcement to the air cathode, preventing it from breaking down or cracking during use. This results in fewer instances of electrode delamination and improved long-term stability.
- Corrosion Resistance: Graphene’s inert nature helps protect metal anodes from corrosion, thus increasing the overall cycle life of the battery. In particular, graphene oxide has been shown to form a protective layer over metal surfaces, improving the lifespan and reducing the degradation of metal-air batteries.
5. Cost Reduction and Scalability
Despite its high performance, graphene is still relatively expensive, which can be a limiting factor in large-scale battery production. However, with advancements in production methods, graphene is becoming more affordable, making its integration into metal-air batteries more cost-effective. Moreover, the scalability of graphene-based materials can lead to significant cost savings in the manufacturing of these batteries.
- Large-Scale Synthesis: New methods of producing graphene oxide and reduced graphene oxide at scale have made it possible to use these materials in larger quantities without significantly increasing the cost of metal-air batteries.
Applications of Graphene-Enhanced Metal-Air Batteries
The integration of graphene into metal-air batteries opens up several promising applications, particularly in fields that require high energy density and long-lasting power solutions. Some notable areas include:
1. Electric Vehicles (EVs)
Metal-air batteries, especially lithium-air batteries, have the potential to offer energy densities far greater than traditional lithium-ion batteries. This could significantly extend the driving range of electric vehicles (EVs), making them more competitive with gasoline-powered vehicles.
- Graphene-enhanced lithium-air batteries could potentially increase the energy density of EV batteries, enabling longer travel distances on a single charge and reducing the need for frequent recharging.
2. Portable Electronics
With the growing demand for longer-lasting portable electronics such as smartphones, laptops, and wearable devices, graphene-enhanced metal-air batteries could offer substantial improvements over conventional rechargeable batteries.
- Smaller, lightweight batteries with high energy density could lead to thinner, longer-lasting electronics that require less frequent charging, enhancing the user experience.
3. Grid-Scale Energy Storage
Metal-air batteries are considered potential candidates for large-scale energy storage solutions, especially for renewable energy systems like wind and solar. The ability to store large amounts of energy and release it efficiently is crucial for stabilizing energy grids.
- Graphene can help metal-air batteries meet the demands of grid-scale storage by improving their efficiency and longevity, making them a viable option for sustainable energy systems.
4. Medical Devices
Long-lasting, lightweight batteries are crucial for medical implants and wearable health devices. Metal-air batteries enhanced with graphene could offer the required energy capacity while reducing the size and weight of batteries used in medical technologies.
Conclusion
Graphene is playing a pivotal role in addressing the challenges associated with metal-air batteries, such as poor conductivity, instability, and limited cycle life. By enhancing the conductivity of the air cathode, improving the stability of the metal anode, and increasing energy density, graphene is helping to unlock the potential of these batteries in applications ranging from electric vehicles to large-scale energy storage. As graphene production becomes more scalable and cost-effective, its integration into metal-air battery technologies promises to drive the development of more efficient, long-lasting, and high-capacity energy storage solutions for the future.